- 503Pharma
- Posts
- 503Pharma Compliance Intelligence
503Pharma Compliance Intelligence
GLP-1s Dispensed April 9. Failed Testing April 28. Patients Were Never Called.
Are your syringe kits costing more than they should?
Most 503A pharmacies assemble syringe kits in-house — sourcing syringes, alcohol pads, packaging them per order. It works, but it adds up in staff time and shelf space.
We're exploring a pre-packaged syringe kit option through our GPO to see if group pricing makes sense for members.
We need a quick read on what you're currently using.
🚨 BREAKING: FDA Expected to Lift Restrictions on 14+ Peptides
Today, the New York Times reported that the FDA is moving toward allowing compounding pharmacies to produce more than a dozen injectable peptides previously banned due to safety concerns.
This follows HHS Secretary Robert F. Kennedy Jr.'s February announcement that approximately 14 of the 19 peptides on the FDA's Category 2 restricted list would be moved to Category 1 — restoring legal compounding access with a physician prescription. Peptides expected to return include BPC-157, Thymosin Alpha-1, AOD-9604, GHK-Cu, and others used for tissue healing, immune support, and metabolic health.
📋 THIS WEEK'S CASE
Welcome back to 503Pharma Compliance Intelligence.
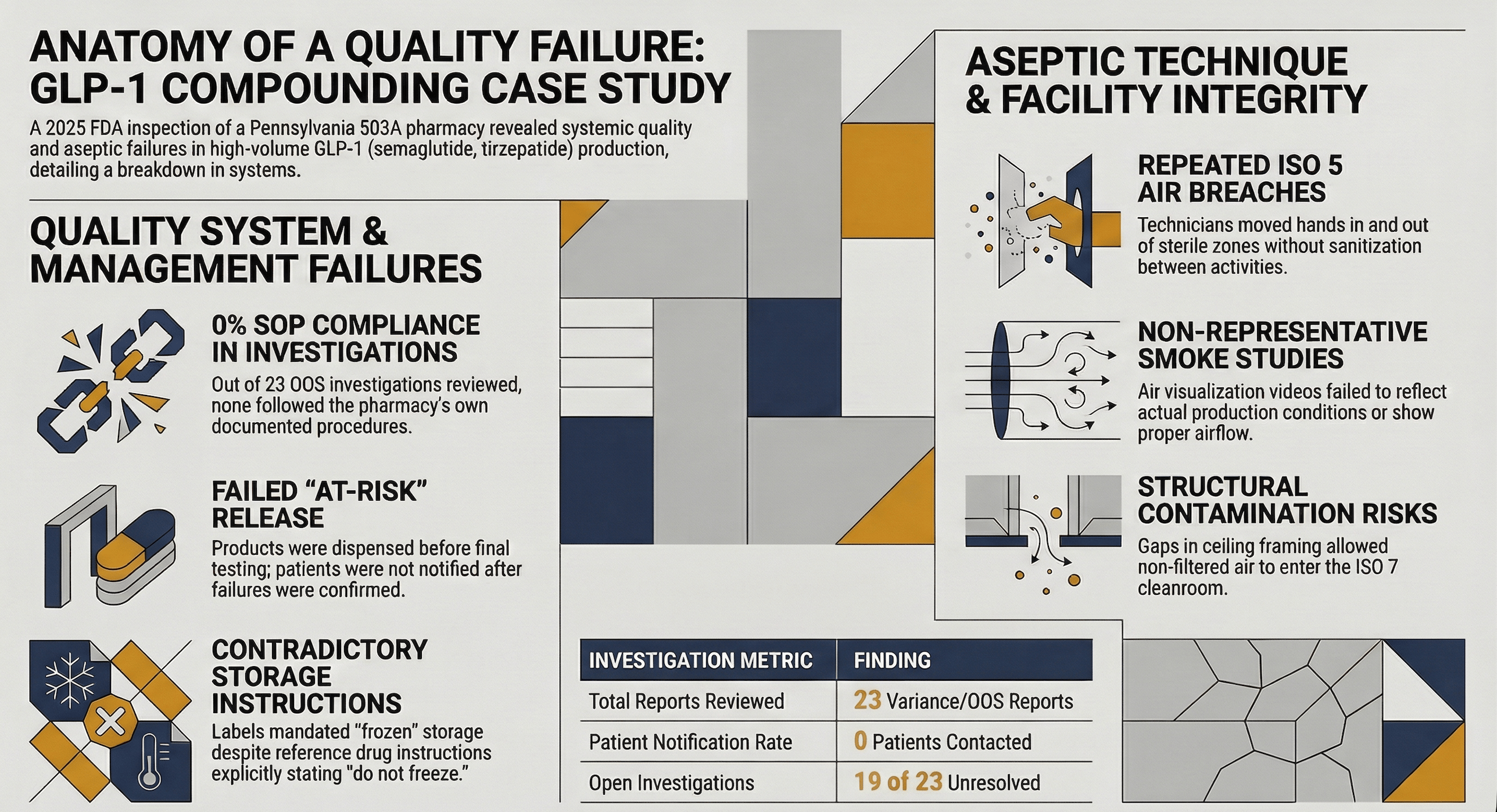
This week: a June 2025 FDA inspection of a Pennsylvania 503A compounding GLP-1 injectables. The pharmacy released product before test results came back. The results failed. Patients weren't notified.
The violations? Observable aseptic technique breaks, smoke studies that didn't represent reality, and 23 OOS investigations — zero of which followed the pharmacy's own procedure.
Let's get into it.
WHAT HAPPENED
This pharmacy was compounding high-volume sterile GLP-1 injectables — semaglutide and tirzepatide formulations. They released product "at risk" before final testing was complete.
Then the test results came back.
Observation 1: Products failed. Patients weren't told.
A tirzepatide lot was compounded on April 7, 2025 and dispensed to patients April 9–12. On April 11, the contract lab flagged a delay — results were "under investigation." On April 28, the lab confirmed the samples failed.
The FDA reviewed 23 variance/OOS investigation reports from November 2024 through May 2025. None followed the pharmacy's own SOP. None documented contacting affected patients. Nineteen of 23 remained open at the time of inspection.
Observation 2: Storage conditions contradicted the reference drug.
Labels on semaglutide and tirzepatide formulations stated "store frozen" — but no stability studies supported frozen storage. The approved drug labeling explicitly states: "Do not freeze and do not use if it has been frozen."
Observation 3: Sterile materials exposed to lower-than-ISO-5 air.
A technician opened pre-sterilized vials outside the ISO 5 area, then used them to fill a production lot. Another technician repeatedly rested their wrist on the front vent of the BSC during filling — moving their hand outside ISO 5 air, then back inside without sanitization. Over and over.
Observation 4: First air blocked during aseptic manipulations.
A technician reached across uncovered vials to grab a filling needle. Paper packaging blocked first-pass air over open vials. Basic aseptic principles — violated in real time.
Observation 5: Smoke studies didn't represent actual operations.
The FDA reviewed the pharmacy's air visualization videos. None showed whether air was entering from ISO 7 into the ISO 5 zone. None represented actual production conditions. Videos showed stagnant air on the deck, no first-pass air over vials, and turbulence with backflow.
Observation 6: Facility surfaces couldn't be properly cleaned.
Ceiling tiles in the ISO 7 cleanroom were recessed and porous — difficult to clean, easy to harbor contaminants. Gaps in ceiling framing allowed non-HEPA-filtered air from the plenum to enter the cleanroom. Drywall walls with epoxy paint left unsmooth surfaces.
WHY THIS MATTERS FOR ALL OF US
1. "At-risk release" without follow-through isn't a quality system — it's just release.
Releasing sterile products before final testing can be defensible. But only if you have rigorous tracking, communication protocols, and notification systems. Without those controls, you're gambling with patient safety and calling it a process.
2. Zero out of 23 investigations followed their own SOP.
Having written procedures creates an expectation of compliance. When an FDA investigator finds that none of your investigations followed your own documented process, it demonstrates not just execution failure — but management failure.
3. Aseptic technique is observable.
The FDA didn't rely on test results alone. They watched technicians work. They saw hands leave ISO 5 air and return without sanitization. They saw first air blocked by packaging. They saw vials opened in the wrong zone. Your technique is your sterility assurance — and it's visible to anyone watching.
4. Smoke studies must represent reality.
Air visualization isn't a checkbox. If your smoke study shows a second person assisting but your actual production uses one person, you haven't validated your process. If your videos don't show airflow where you actually place materials, you don't know what's happening in your critical zone.
🛡️ BUILDING SYSTEMS THAT CATCH THIS
"At-risk release" requires bulletproof tracking — because when results fail, every minute counts. And aseptic technique isn't a training topic. It's a daily observable behavior.
Here's how CompoundLearn helps you stay ahead:
Training Modules Build competency in aseptic processing with C-1: Garbing & Hygiene in Practice and C-3: Compounding Workflows — so your team understands first air, proper technique, and why hands don't leave the ISO 5 zone mid-operation. Add DP-4: Quality Assurance Program for OOS response and patient notification protocols.
SOP Library Access ready-to-use procedures for at-risk release tracking, OOS investigations, patient notification workflows, and smoke study protocols. Don't reinvent the wheel — customize proven templates.
Competencies Assign and track role-specific qualifications. Ensure the person releasing product understands what "at-risk" actually requires. Document who's trained on what.
Templates Generate OOS investigation forms, patient notification logs, and smoke study documentation. Audit-ready records that take seconds to create.
Assessments Verify that your team can identify aseptic technique breaks and respond appropriately. Document the results automatically.
One unified system for training, documentation, and compliance.
A 483 for products you already shipped. Or a system that ensures you act before patients are affected.
✅ THIS WEEK'S ACTION ITEM
Review your at-risk release process. If you dispense before final testing:
Is there a documented tracking system for every lot?
Is there a defined notification protocol if results fail?
Can you reach every patient within 24 hours of a confirmed failure?
If you can't answer yes to all three, you don't have an at-risk release program. You have a liability.
Reply and tell us: Was this useful? What would make it better?
✉️ That’s your 503Pharma Intel Brief. Our mission is to keep compounding professionals informed, prepared, and ahead of the curve.
