- 503Pharma
- Posts
- 503Pharma Weekly Compounding Pharmacy Roundup
503Pharma Weekly Compounding Pharmacy Roundup
Your trusted source for compounding pharmacy education, news and insights.
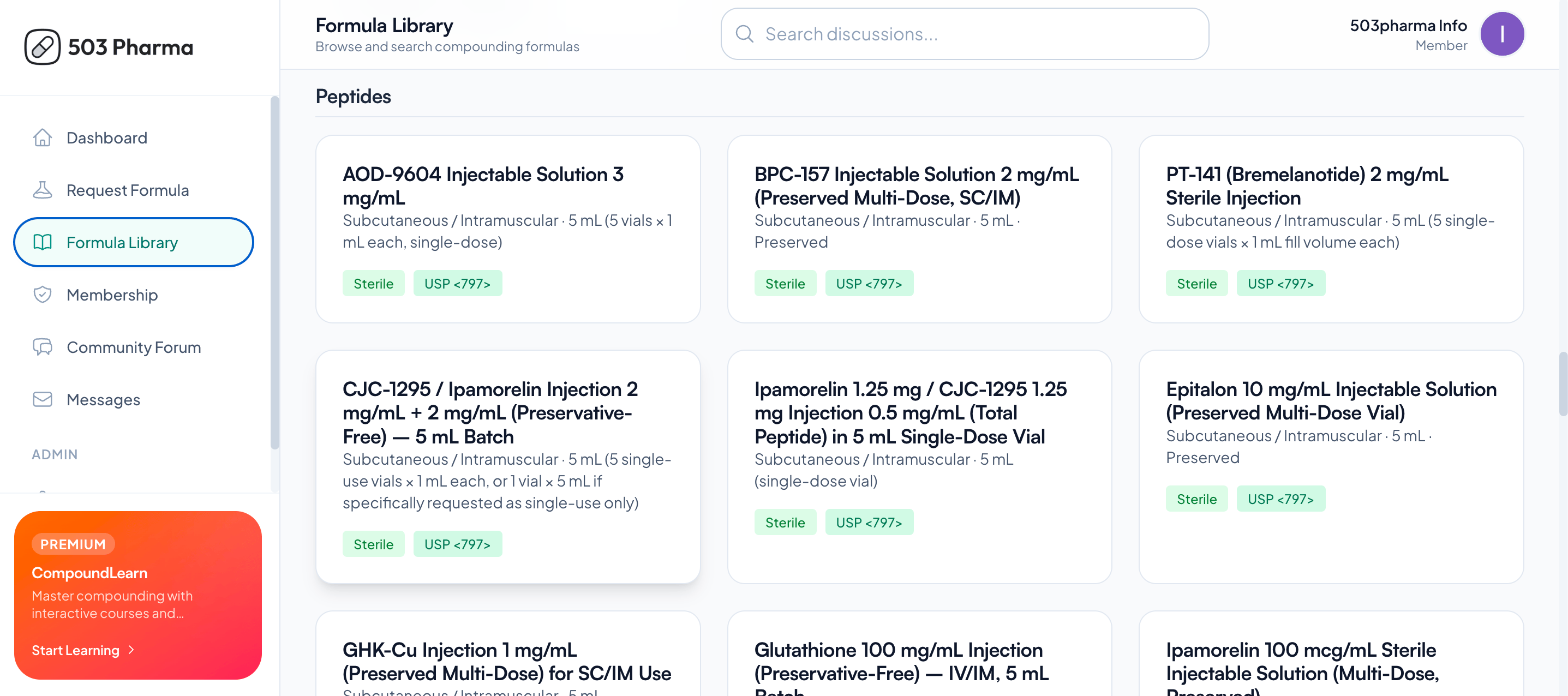
What's Next After GLP-1? 90+ Formulas to Build a Resilient Formulary
The 503Pharma Formula Library just launched at app.503pharma.com. Ready-to-use formulations for sterile and non-sterile compounding, built to current USP 795/797 standards, with BUD assignments included. Updated as guidance changes.
90+ formulations across sterile and non-sterile categories
BUD assignments aligned to USP 795 and 797
Custom formulation requests
Regularly updated as FDA and USP guidance evolves
The members area is free to join. Formula access is a paid feature.
This Week's Headlines
Market
Lilly's Foundayo Hits U.S. Pharmacies
Lilly's oral GLP-1 pill Foundayo (orforglipron) is now at retail pharmacies and telehealth — $149/month self-pay, no food or water restrictions, any time of day. Approved in 50 days under FDA's fast-track CNPV program, the fastest NME approval since 2002. Second oral GLP-1 after the Wegovy pill in December. Two branded oral options at consumer prices accelerates the shift away from compounded GLP-1 injectables.
Industry
Noom Acquires Tailor Made Compounding
Noom bought Tailor Made Compounding — 503A pharmacy, 46 states, 40,000+ sq ft Kentucky facility, 400+ clinic relationships. Formulary: sermorelin, GHK-Cu, NAD+, oxytocin, HRT. Not a GLP-1 play. A vertical integration bet on preventive care through compliant 503A infrastructure.
Regulatory
FDA Fast-Track Approvals Are Compressing the Compounding Window
FDA's CNPV program: five approvals in 10 months — Foundayo in 50 days, Wegovy HD in 54, a lung cancer drug in 44. Standard review is 10-12 months. Faster branded approvals shrink the supply-gap argument for compounding copies and strengthen the case for differentiated formulations.
Research Desk
Peptide Investigational New Drug Pipeline
BPC-157 hamstring trial (Phase 2, 120 participants) and TB-500 cardiovascular trial (Phase 1/2, 80 participants) both actively recruiting.
ReGenTree's Phase 3 RGN-259 trial (thymosin beta 4 ophthalmic solution) hits primary completion this month across 36 sites — potential sterile ophthalmic compounding opportunity if the branded product stalls. Every major compounding peptide is now in a formal IND pathway.
GLP-1 Indications Expanding
UF Phase 2 started April 7 for endometrioid intraepithelial neoplasia.
Weill Cornell Phase 4 tirzepatide in postmenopausal breast cancer survivors.
UTSW semaglutide for subclinical Cushing's.
University of Colorado Phase 2/3 testing semaglutide combined with HRT and antihypertensives for menopausal transition — exactly the multi-drug personalized protocol where 503A pharmacies add unique value.
GLP-1 Delivery Innovation
Vivani Medical's semaglutide subcutaneous implant enters Phase 1 this month vs. weekly Wegovy.
Shanghai World Leader's semaglutide nasal spray already recruiting Phase 1. New delivery forms could shift prescribing away from injectables entirely.
Low-Dose Naltrexone (LDN) Evidence Base Growing
UAB dose-finding trial for LDN in ME/CFS (1.5–6.0mg) launches July. University of Arkansas Phase 2 testing LDN for fatigue in prostate cancer patients on ADT. Johns Hopkins Phase 2 evaluating semaglutide + naltrexone for alcohol use disorder — a combo compounders are uniquely positioned to provide.
Intel Briefs
Wegovy HD (7.2mg) Approved March 19
Higher-dose semaglutide injectable approved in 54 days via CNPV. Novo simultaneously received a Warning Letter on March 5 for systemic adverse-event reporting failures — speed to approval doesn't exempt you from post-market compliance. Pharmaceutical Technology
3.1M Eye Drops Recalled — Sterility Failure
KC Pharmaceuticals recalled 3.1M bottles (CVS, Walgreens, Publix, Rite Aid) — Class II, lack of sterility assurance. FDA had flagged the same contamination issues in a 2023 warning letter. Sterile manufacturing credibility is a differentiator when big players stumble. US News
Rifampin Manufacturing Ceased
FDA confirmed capsule production has halted entirely. TB programs are reaching out to 503B facilities for emergency supply. Compounders with capsule capabilities should secure rifampin API now — expect competition and price spikes. FDA Drug Shortages Database
NEW: 503Pharma Formula Library
We just launched a formula library for compounding pharmacies — 90+ ready-to-use formulations, now live in the members area at app.503pharma.com.
Every formula is built for real compounding workflows: USP 795/797-compliant, sterile and non-sterile coverage, beyond-use dating included, and updated as guidance evolves.
What's inside:
90+ formulations across sterile and non-sterile categories
BUD assignments aligned to USP 795/797
Ready-to-use format — drop into your compounding records directly
Regularly updated as FDA and USP guidance changes
The members area is free to join. Formula access is a paid feature.
CompoundLearn: Training Built for Compounding Pharmacies
CompoundLearn simplifies USP compliance training so your team can stay inspection-ready without the spreadsheets and paper trails. From onboarding new staff to competency testing, CompoundLearn automates the entire training lifecycle — so you can focus on what matters most: patient safety.
Full access to 25 training modules and 77 lessons covering critical USP 795, 797, and 800 topics
Role-based assignments — assign and track the right training curriculum to designated persons/pharmacists-in-charge, compounders, and trainers
Quizzes and Downloadable certificates that prove that your team knows the material
23+ Ready-Made Compliance Templates — Downloadable cleaning logs, sampling records, labeling checklists, and more — ready to use out of the box
Custom Module Creation — Build pharmacy-specific training for your unique protocols and SOPs
Team & Role Management — Assign training by role (designated person, compounder, technician) and track progress across your entire organization
✉️ That’s your 503Pharma weekly digest. Our mission is to keep compounding professionals informed, prepared, and ahead of the curve.